GSK
See the following -
"Industry Will Not Support Open-Ended Science, So Govt Must"
On December 11, News18 exposed how India’s clinical trials and drug discovery process is skewed towards diseases like cancer while ignoring the top killers of the country like TB, diarrhea and Kala Azar. Responding to that, Dr. Soumya Swaminathan, Director-General of the Indian Council for Medical Research (ICMR) told News18 there was a need to support indigenous research in India. In this interview to News18’s Aradhna Wal, Dr Swaminathan says India needs a 10-year vision on drug research...
- Login to post comments
Approval Of A Coronavirus Vaccine Would Be Just The Beginning - Huge Production Challenges Could Cause Long Delays
 The race for a SARS-CoV-2 vaccine is well underway. It's tempting to assume that once the first vaccine is approved for human use, all the problems of this pandemic will be immediately solved. Unfortunately, that is not exactly the case. Developing a new vaccine is only the first part of the complex journey that's supposed to end with a return to some sort of normal life. Producing hundreds of millions of vaccines for the U.S. - and billions for the world as a whole - will be no small feat. There are many technical and economic challenges that will need to be overcome somehow to produce millions of vaccines as fast as possible.
The race for a SARS-CoV-2 vaccine is well underway. It's tempting to assume that once the first vaccine is approved for human use, all the problems of this pandemic will be immediately solved. Unfortunately, that is not exactly the case. Developing a new vaccine is only the first part of the complex journey that's supposed to end with a return to some sort of normal life. Producing hundreds of millions of vaccines for the U.S. - and billions for the world as a whole - will be no small feat. There are many technical and economic challenges that will need to be overcome somehow to produce millions of vaccines as fast as possible.
- Login to post comments
Drug Giant Glaxo Pleads Guilty, Fined $3B for Drug Marketing
Drug giant GlaxoSmithKline will plead guilty and pay $3 billion to resolve federal criminal and civil inquiries arising from the company's illegal promotion of some of its products, its failure to report safety data and alleged false price reporting, the Justice Department announced Monday. Read More »
- Login to post comments
EU Agency Lifts Lid on Drug Data Secrets
Europe's medicines regulator, criticised in the past for excessive secrecy, is opening its data vaults to systematic scrutiny in a move that will let independent researchers trawl through millions of pages of clinical trial information. Read More »
- Login to post comments
European Bioinformatics Institute (EMBL-EBI) ChEMBL 20 incorporates the Pistoia Alliance’s HELM annotation
 The European Bioinformatics Institute (EMBL-EBI) has released version 20 of ChEMBL, the database of compound bioactivity data and drug targets. ChEMBL now incorporates the Hierarchical Editing Language for Macromolecules (HELM), the macromolecular representation standard recently released by the Pistoia Alliance.
The European Bioinformatics Institute (EMBL-EBI) has released version 20 of ChEMBL, the database of compound bioactivity data and drug targets. ChEMBL now incorporates the Hierarchical Editing Language for Macromolecules (HELM), the macromolecular representation standard recently released by the Pistoia Alliance.
- Login to post comments
Giant GSK Settlement Provides Reminder of the Pervasiveness of Stealth Marketing
The latest and biggest legal settlement involving health care to hit the news, that of GlaxoSmithKline (GSK) and the US government, has many familiar elements. [These documents] provide good documentation about how pervasive systematic, deceptive stealth marketing campaigns have become in health care. Read More »
- Login to post comments
King's College Accelerates Synthetic Brain 3D Image Creation Using Open Source AI Models and Software Powered by Cambridge-1 Supercomputer
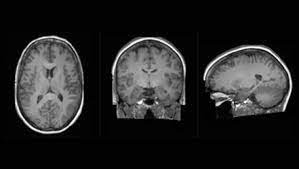 King College London, along with partner hospitals and university collaborators, unveiled new details today about one of the first projects on Cambridge-1, the United Kingdom's most powerful supercomputer. The Synthetic Brain Project is focused on building deep learning models that can synthesize artificial 3D MRI images of human brains. These models can help scientists understand what a human brain looks like across a variety of ages, genders, and diseases. The AI models were developed by King's and NVIDIA data scientists and engineers as part of The London Medical Imaging & AI Centre for Value Based Healthcare research funded by UK Research and Innovation and a Wellcome Flagship Programme (in collaboration with University College London).
King College London, along with partner hospitals and university collaborators, unveiled new details today about one of the first projects on Cambridge-1, the United Kingdom's most powerful supercomputer. The Synthetic Brain Project is focused on building deep learning models that can synthesize artificial 3D MRI images of human brains. These models can help scientists understand what a human brain looks like across a variety of ages, genders, and diseases. The AI models were developed by King's and NVIDIA data scientists and engineers as part of The London Medical Imaging & AI Centre for Value Based Healthcare research funded by UK Research and Innovation and a Wellcome Flagship Programme (in collaboration with University College London).
- Login to post comments
Open Source Drug Discovery Test A Success
 In what is being called the first-ever test of open-source drug-discovery, researchers from around the world have successfully identified compounds to pursue in treating and preventing parasite-borne illnesses such as malaria as well as cancer...One-third of the labs reported their results in a paper published today in PLOS Pathogens, "Open source drug discovery with the Malaria Box compound collection for neglected diseases and beyond." The results have ignited more a dozen drug-development projects for a variety of diseases. "The trial was successful not only in identifying compounds to pursue for anti-malarials, but it also identified compounds to treat other parasites and cancer," said lead author Wesley Van Voorhis.
In what is being called the first-ever test of open-source drug-discovery, researchers from around the world have successfully identified compounds to pursue in treating and preventing parasite-borne illnesses such as malaria as well as cancer...One-third of the labs reported their results in a paper published today in PLOS Pathogens, "Open source drug discovery with the Malaria Box compound collection for neglected diseases and beyond." The results have ignited more a dozen drug-development projects for a variety of diseases. "The trial was successful not only in identifying compounds to pursue for anti-malarials, but it also identified compounds to treat other parasites and cancer," said lead author Wesley Van Voorhis.
- Login to post comments
Open Targets: New Name, New Data
 Following the successful launch of its Target Validation platform at the end of 2015, the Centre for Therapeutic Target Validation has released its first open experimental datasets. Now renamed Open Targets, the pioneering public–private initiative remains committed to speeding up the discovery of new medicines. Open Targets projects use genome-scale experiments and analysis to provide evidence on the biological validity of therapeutic targets – and to glean insights into the likely effectiveness of pharmacological intervention on these targets...
Following the successful launch of its Target Validation platform at the end of 2015, the Centre for Therapeutic Target Validation has released its first open experimental datasets. Now renamed Open Targets, the pioneering public–private initiative remains committed to speeding up the discovery of new medicines. Open Targets projects use genome-scale experiments and analysis to provide evidence on the biological validity of therapeutic targets – and to glean insights into the likely effectiveness of pharmacological intervention on these targets...
- Login to post comments